|
This leaves an opening for one single bond to form. It requires just one more electron to be full. It has a single electron in the 1s orbital. Let’s take a quick detour to review electron configuration with a focus on valence electrons, as they are the ones that actually participate in the bond.įeeling rusty? Click to review my Electron Configuration + Shortcut videos. In order to create a covalent bond (video), each participating atom must have an orbital ‘opening’ (think: an empty space) to receive and interact with the other atom's electrons. With covalent bonds being the strongest and most prevalent. Simply put, molecules are made up of connected atoms,Ītoms are connected through different types of bonds, Molecules are everywhere! How do they form? In addition to undergrad organic chemistry, this topic is critical for exams like the MCAT, GAMSAT, DAT and more. Sp³ d and sp³ d² Hybridization – An Overview. 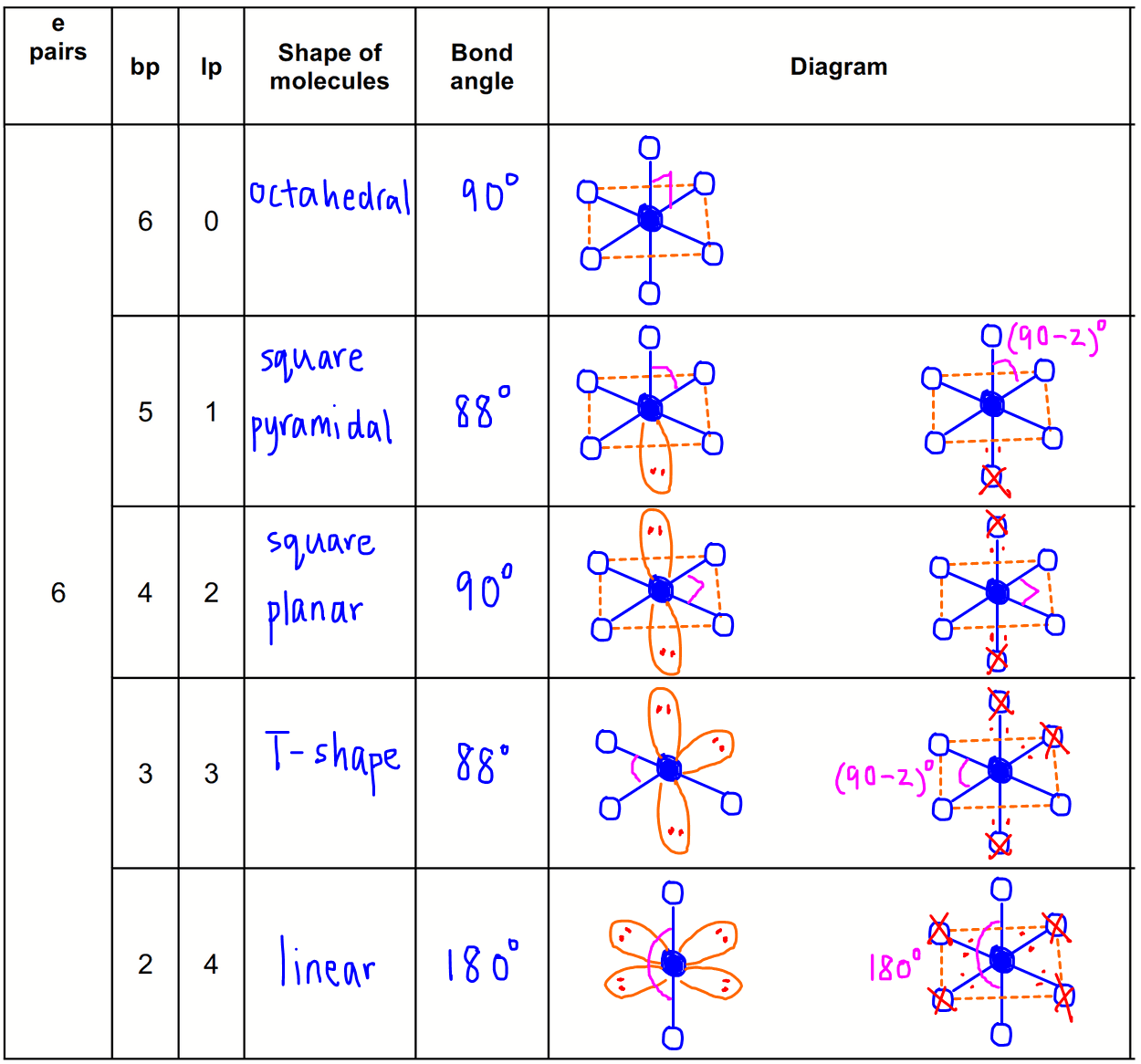
Sp Hybridization, Bond Angle and Geometry.  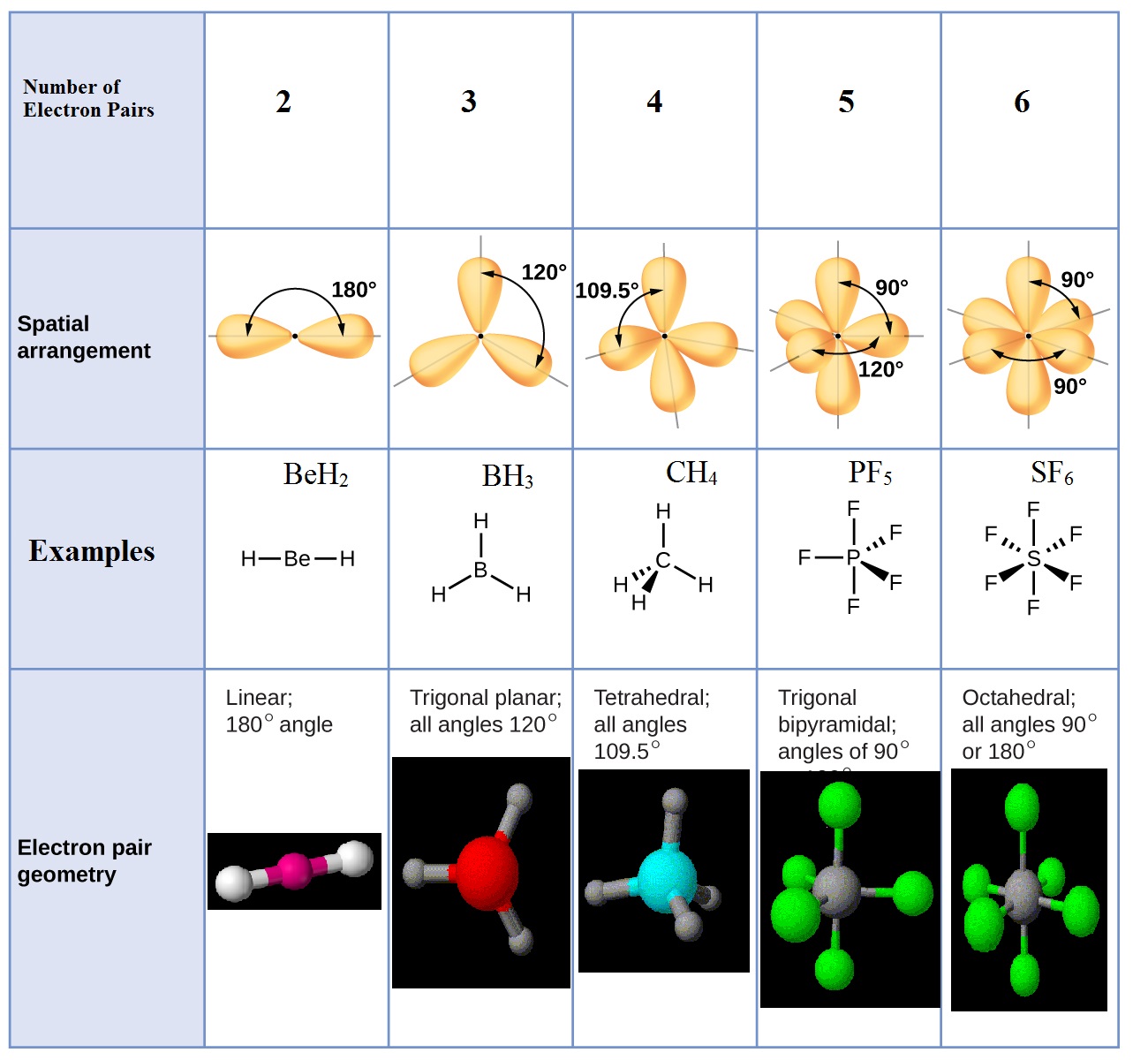
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
